|
6/4/2023 0 Comments Pfizer vaccine 2nd dose timing (“Pfizer”)) in the amount of €3,961.3 million was received from our collaboration partner as of April 14, 2023. Subsequent to the end of the reporting period, the payment settling BioNTech’s gross profit share for the fourth quarter of 2022 (as defined by the contract with Pfizer, Inc. Net profit was €502.2 million for the three months ended March 31, 2023, compared to €3,698.8 million for the comparative prior year period.Ĭash and cash equivalents as well as security investments were €12,143.9 million and €671.9 million, respectively, as of March 31, 2023. The derived annual effective income tax rate for the three months ended March 31, 2023, was 29.0% which is expected to decrease over the 2023 financial year to be in line with BioNTech’s guidance. Income taxes were accrued in an amount of €205.5 million of tax expenses for the three months ended March 31, 2023, compared to €1,319.3 million of tax expenses for the comparative prior year period. The change was mainly due to increased expenses for IT, purchased external services, as well as an increase in headcount. General and administrative expenses were €119.4 million for the three months ended March 31, 2023, compared to €90.8 million for the comparative prior year period. The increase was further driven by an increased headcount. The change was mainly due to higher expenses incurred from progressing the clinical studies for pipeline candidates. Research and development expenses were €334.0 million for the three months ended March 31, 2023, compared to €285.8 million for the comparative prior year period. The change was mainly due to decreasing sales from BioNTech’s COVID-19 vaccine revenues. The change was mainly due to lower commercial revenues from the supply and sales of the Company’s COVID-19 vaccines worldwide.Ĭost of sales were €96.0 million for the three months ended March 31, 2023, compared to €1,294.1 million for the comparative prior year period. Total revenues reported were €1,277.0 million 2 for the three months ended March 31, 2023, compared to €6,374.6 million 2 for the comparative prior year period. For the remainder of 2023, we are focused on advancing our disruptive platforms against solid tumors and accelerating clinical programs in infectious diseases of high global need.”įinancial Review for the First Quarter 2023 in millions €, except per share data We are also making progress in advancing our next generation COVID-19 vaccine candidate while we stand prepared for variant adaptation in case of public health need. “We are taking significant steps in this direction as we prepare to initiate our first Phase 3 clinical trial in oncology for the novel anti-CTLA-4 antibody in NSCLC patients who have progressed after PD-1/PD-L1 treatment, a patient population with high medical need. Ugur Sahin, M.D., CEO and Co-Founder of BioNTech. 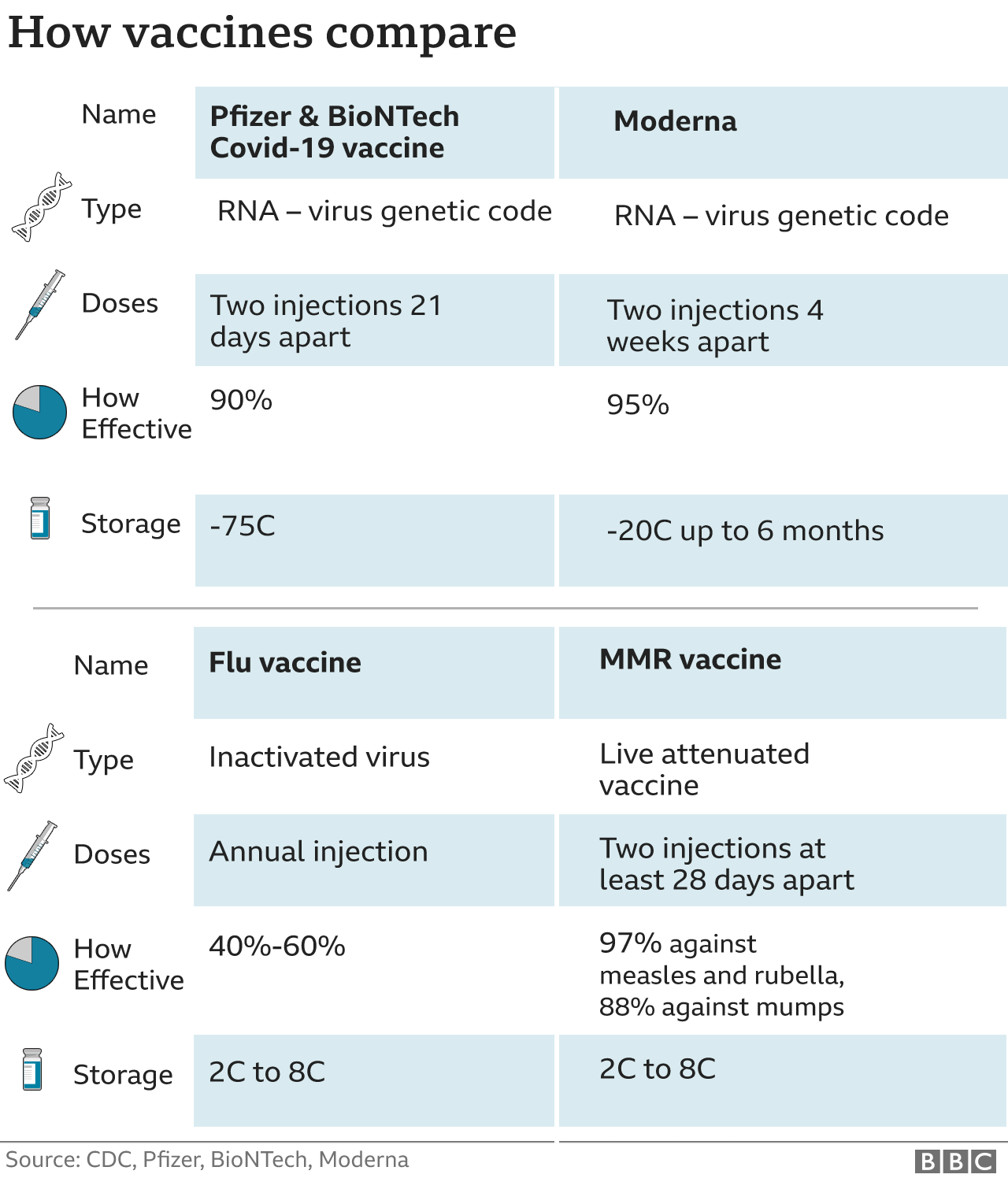 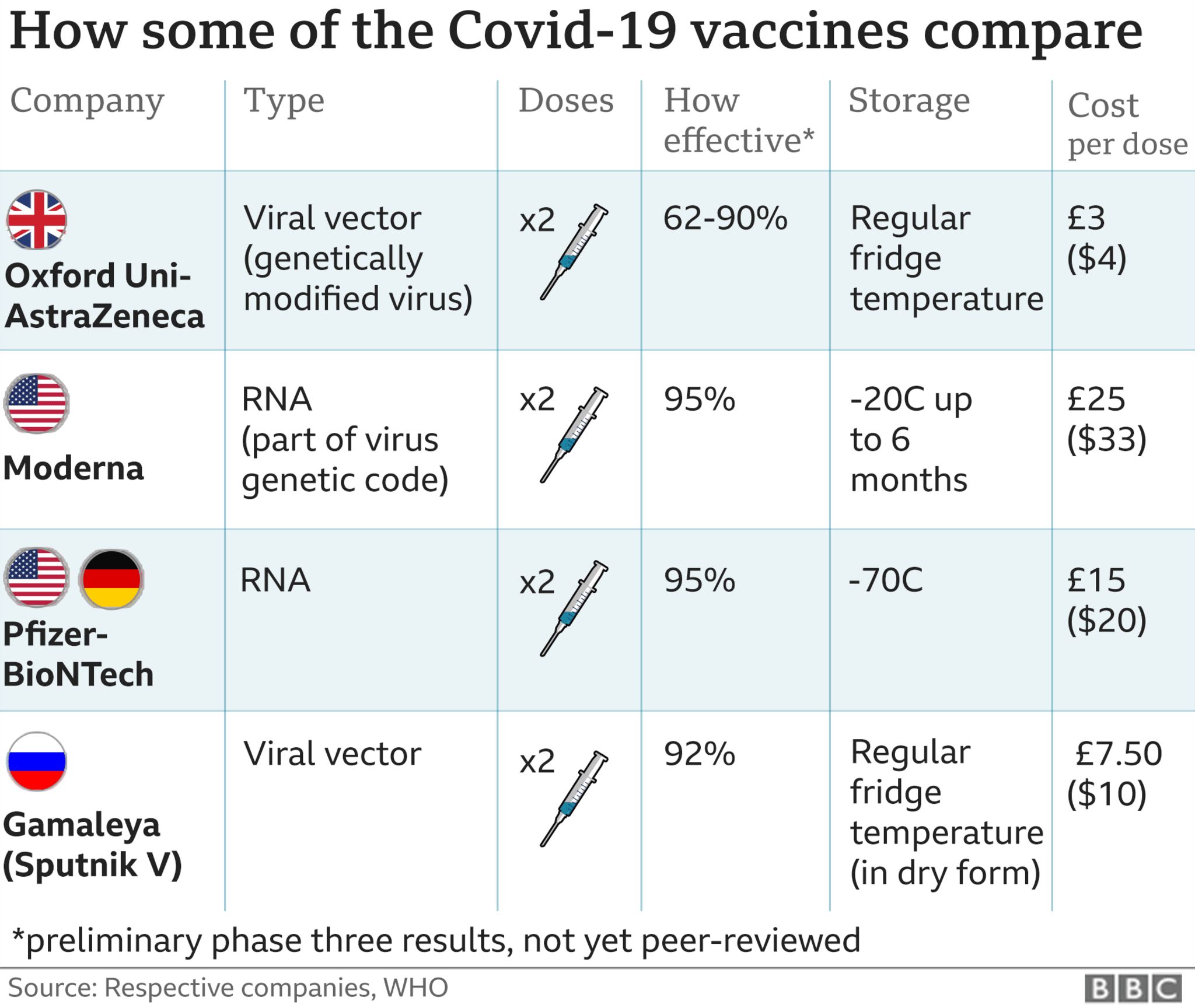
These programs are strategically aligned with our vision to provide meaningful therapeutic benefits for patients with solid tumors along the entire treatment journey,” said Prof. “In the first quarter of 2023, we expanded our toolkit of cutting-edge technologies to new modalities and added a novel immune checkpoint inhibitor candidate targeting CTLA-4 and two investigational antibody-drug conjugates to our arsenal against cancer. MAINZ, Germany, (GLOBE NEWSWIRE) - BioNTech SE (Nasdaq: BNTX, “BioNTech” or the “Company”) today reported financial results for the three months ended March 31, 2023, and provided an update on its corporate progress. First quarter 1 revenues of €1.3 billion 2, net profit of €0.5 billion and fully diluted earnings per share of €2.05 ($2.20 3)Ĭonference call and webcast scheduled for May 8, 2023, at 8:00 am EDT (2:00 pm CEST).Reiterates BioNTech COVID-19 vaccine revenue guidance of approximately €5 billion in 2023.Broadened clinical-stage infectious disease vaccine pipeline with the start of a First-in-Human clinical trial for the first mRNA-based Tuberculosis vaccine candidates.Presenting clinical data on antibody candidate BNT316 (ONC-392), ADC candidate BNT323 (DB-1303) and CAR-T candidate BNT211 at the 2023 American Society of Clinical Oncology Annual Meeting.Added new class of precision therapeutics to clinical-stage oncology portfolio, with next-generation Antibody-Drug Conjugate (ADC) candidates.BioNTech and partner OncoC4 plan to start a Phase 3 clinical trial evaluating anti-CTLA-4 antibody BNT316 (ONC-392) as monotherapy in NSCLC patients who progress after PD-1/PD-L1 treatment.COVID-19 vaccine franchise focused on vaccine adaptation readiness ahead of the fall season and advancing next generation vaccine candidates and combinations. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
